
May 3, 2021
UCalgary researchers discover link between prenatal stress and altered brain development
Neuroimmune cells overwhelmed by stress are now known to be key contributors underpinning neurodevelopmental disorders. UCalgary scientists, working with mice, found a relationship between prenatal stress and disruptions in brain development, especially in male offspring.
University of Calgary scientists Dr. Jessica Rosin, PhD'15, and Dr. Deborah Kurrasch, PhD, a member of the Alberta Children’s Hospital Research Institute (ACHRI) at the Cumming School of Medicine (CSM) led the research published in Developmental Cell.
“This paper starts to provide key linkages into how the maternal environment influences the developing brain and also underscores differences in its responses between males and females,” notes Rosin, the lead investigator. Neurodevelopmental disorders are more prominent in boys in some conditions such as autism spectrum disorder (ASD) and attention-deficit/hyperactivity disorders (ADHD). This study in mice may help explain why that happens.
To start, the scientists studied the specific roles of these neural immune cells, called microglia, in the developing brain. They quickly focused on a cluster of microglia in the hypothalamus that appeared to be sensing the maternal environment and reacting to it.
“Microglia appear to be an underappreciated fetal brain sensor, responsible for recognizing adverse cues from the mum and deciding whether to act as a buffer against the prenatal stressor or to inform neighbouring brain cells to adapt,” says Kurrasch, whose lab is focused on establishing the molecular and cellular links between maternal health and adverse brain development in children.
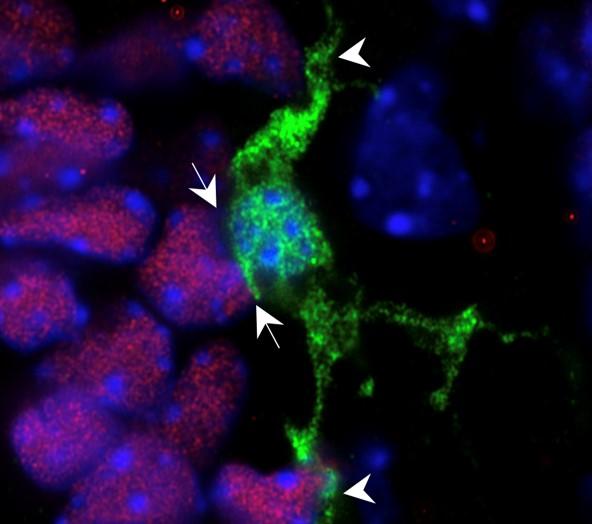
Microglia (green) are in close contact with neural stem cells (pink).
Kurrasch Lab
Exposing pregnant female mice to moderate stress, in this case intermittent cold temperatures, the team found microglia released a tide of inflammatory signals, particularly in male offspring. This elevated neuroimmune signalling, in turn, disrupted hypothalamic development leading to varying levels of dysfunction. The hypothalamus is best known for regulating physiological responses, such as sleep, hunger, body temperature, and thirst — it also plays a key role in social and emotional behaviours.
These findings point to a new function for microglia in the developing brain. “Historically, microglia were thought to be passive bystanders in the embryonic brain; however, emerging studies now show these immune cells to be quite active, with our data demonstrating that their activity can be guided by cues from the maternal environment,” says Kurrasch.
The team is now evaluating why these brain immune cells respond differently in males. “It could be that the female microglia are maturing at a different rate than the male cells, or possibly that female brains have a genetic component that protects them from over-activated microglia,” suggests Rosin.
When the scientists examined the differences in microglial response to prenatal stress in male and female brains, they were surprised to discover 738 downregulated genes in male microglia and only 70 genes downregulated in female microglia. "This may be one clue as to why male brains are more robustly impacted by stress than their female counterparts" says Rosin.
The team also showed that the developmental changes brought on by the prenatal stress had long-term effects. The scientists found that prenatally stressed males displayed deficits in social behaviours, further suggesting that microglia sense the maternal environment and induce changes in the developing brain.
“It is important to understand the role of microglia in the embryonic brain and its relationship to prenatal stress,” says Kurrasch. “With further understanding, we may find a vulnerable pathway for interventions that reduce these insults on the developing brain and potentially improve outcomes for neurodevelopmental disorders.
The research was conducted in collaboration with Dr Jeff Biernaskie, PhD, and Sarthak Sinha, MD-PhD student, and was supported by a Canadian Institutes of Health Research (CIHR) Operating Grant. Jessica Rosin was supported by CIHR, ACHRI and Alberta Innovates Postdoctoral Fellowships. The Kurrasch Lab is supported by the community through the Alberta Children’s Hospital Foundation and Campus Alberta Neuroscience.
Deborah Kurrasch is a professor in the departments of Medical Genetics and Biochemistry & Molecular Biology at the Cumming School of Medicine. She is a member of ACHRI, the Owerko Centre, and Hotchkiss Brain Institute (HBI).
Rosin is a postdoctoral fellow with ACHRI in the Department of Medical Genetics.
Child Health and Wellness
The University of Calgary is driving science and innovation to transform the health and well-being of children and families. Led by the Alberta Children’s Hospital Research Institute, top scientists across the campus are partnering with Alberta Health Services, the Alberta Children’s Hospital Foundation, and our community to create a better future for children through research.






